What is reverse osmosis and how is it used in water treatment?
Reverse osmosis (RO) is a vital part of the water treatment process where the feed water passes through a system that removes impurities & other contaminants to make it pure.
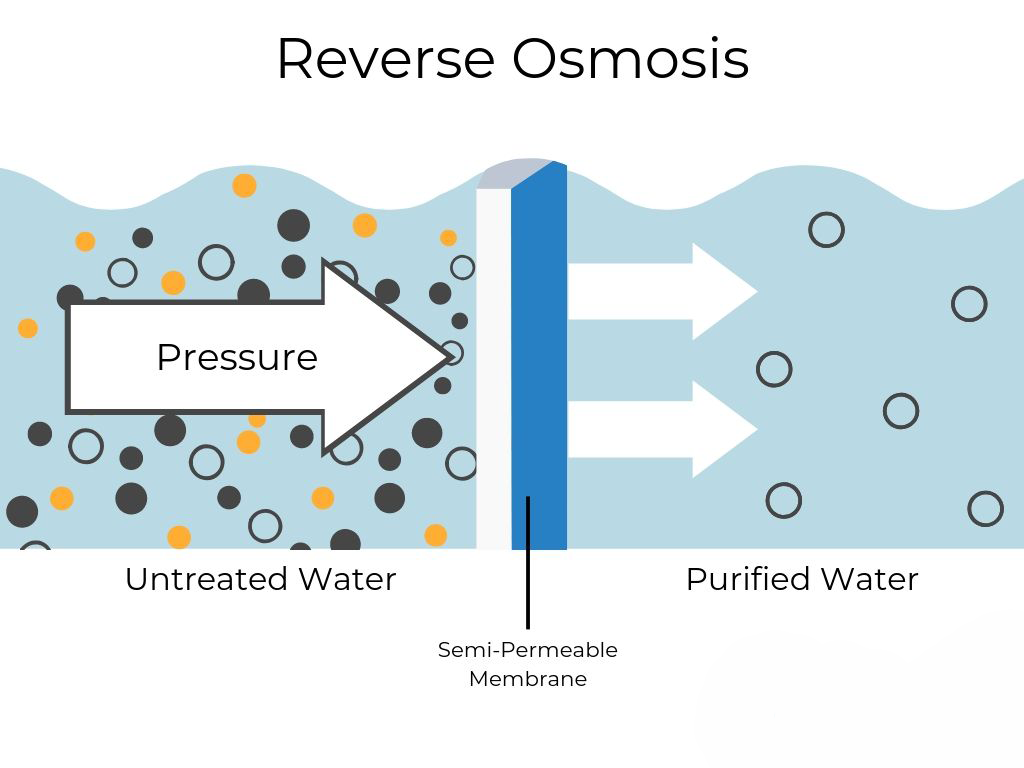
RO systems work by forcing raw water through a semipermeable membrane (at high pressure) so that only the water molecules get through to provide clean drinking water.
The system is more complex than regular filtration systems as it has cutting edge designed membranes that remove the impurities without the use of chemicals, resins or ion exchange beds. Unlike other filtration systems that operate on a low-pressure water system, RO requires a higher pressure pump to drive the water through the membranes in order for it to remove more impurities than other filtration options.
Typical industrial RO’s can remove 97% of the minerals and are 75% efficient (25% waste).
Reverse osmosis is one of the most cost-effective methods of treating boiler feedwater, cooling tower makeup water and industrial process water.
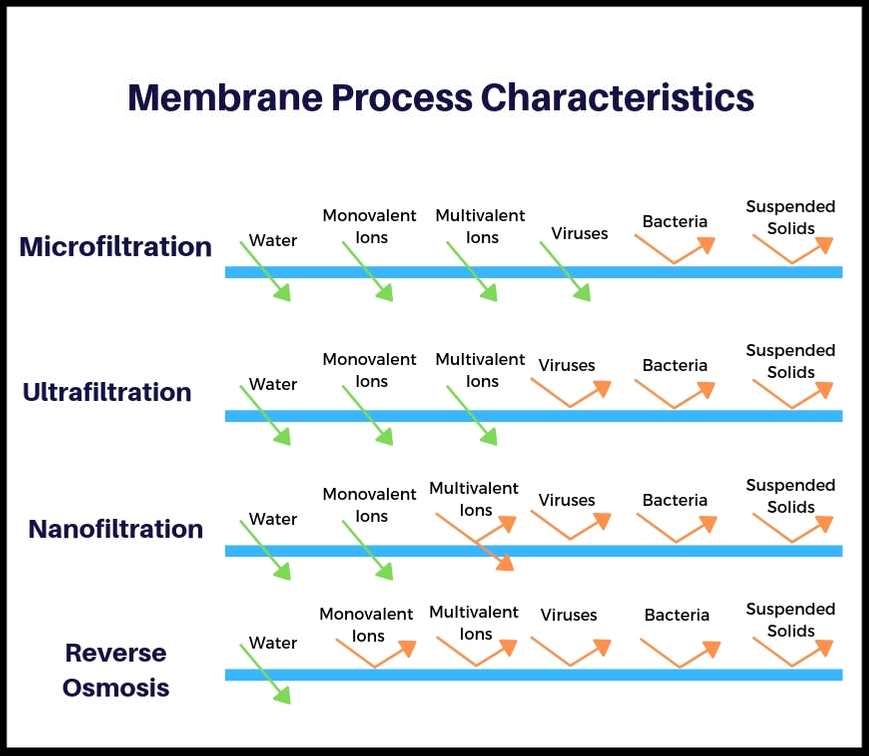
Semi-permeable membrane
The semi-permeable membrane is a membrane that will allow some atoms or molecules to pass but not others.
It has small pores that that allows the passage of water molecules but not the majority of dissolved salts, organics, bacteria and pyrogens.
How does it work?
To understand why reverse osmosis is required it is important to understand osmosis.
Osmosis is a naturally occurring process in nature, where a weaker, less concentrated saline solution will migrate to a strong saline solution with a higher concentration.
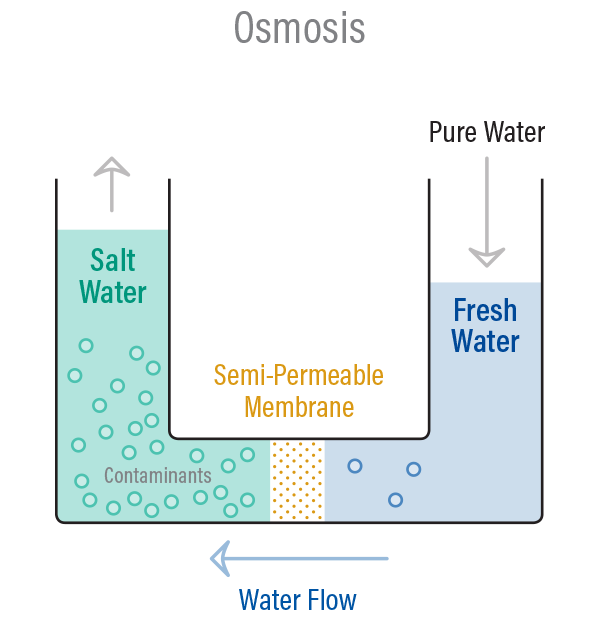
For example, if you had a container full of water with a low salt concentration and another container full of water with a high salt concentration and they were separated by a semi-permeable membrane, then the water with the lower salt concentration would begin to migrate towards the water container with the higher salt concentration.
In osmosis, water becomes more concentrated as it passes through the membrane to obtain equilibrium on both sides, however reverse osmosis, blocks contaminants from entering the less concentrated side of the membrane.
Reverse Osmosis
Reverse Osmosis is basically the process of Osmosis in reverse.
Whereas Osmosis occurs naturally without energy needed, to reverse the process of osmosis you need to apply energy to the more concentrated water.
You need to ‘push’ the water through the reverse osmosis membrane by applying pressure that is greater than the naturally occurring osmotic pressure in order to desalinate the water in the process, this allows the pure water through while holding back a majority of contaminants.
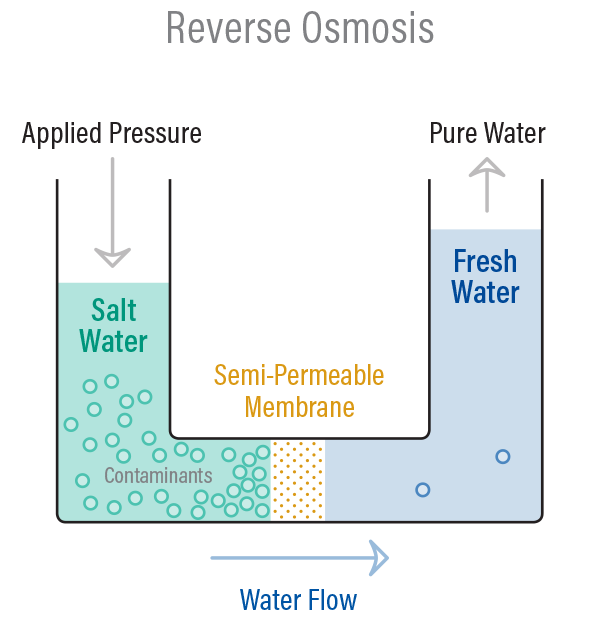
The pressure is produced by a high-pressure pump and leaves almost all (around 95% to 99%) of dissolved salts behind in the reject stream. The amount of pressure required depends on the salt concentration of the feed water. The more concentrated the feed water, the more pressure is required to overcome the osmotic pressure.
The desalinated water that is demineralized or deionized, is called permeate (or product) water. The water stream that carries the concentrated contaminants that did not pass through the RO membrane is called the reject (or concentrate) stream.
What does RO remove?
A reverse osmosis system also includes a sediment and carbon filtration for a broad spectrum of reduction which helps remove more contaminants including:
- Fluoride
- Salt
- Sediment
- Chlorine
- Arsenic
- VOCs
- Herbicides and pesticides
Benefits of the RO system?
A reverse osmosis system is one of the greenest solutions for industrial wastewater and the one of the most extensive methods of filtration, removing 95% to 99% of contaminants making it healthier and safer for consumption. Other benefits include:
- Bad tastes and odours reduced
- More environmentally friendly
- Easy to install and maintain
- More cost efficient
- Not as man health & safety risks
RO doesn’t require the use of harmful chemicals so there is less hazardous wastewater all while capturing impurities allowing you to dispose of them correctly.
When it comes to cost and efficiency, the membranes can last up to 2-3 years depending on varying factors such as the quality and quantity of the water being fed into the RO system. It also means there is limited downtime when it comes to reverse osmosis as membranes can be replaced within hours.
Pre-treatment and design factors?
There are a handful of calculations that are used to judge design type & size of RO systems, including;
- Feed pressure
- Permeate pressure
- Concentrate pressure
- Feed conductivity
- Permeate conductivity
- Feed flow
- Permeate flow
- Temperature
Proper pre – treatment using both mechanical and chemical treatments is also critical ro ensure the RO system works correctly to help prevent fouling, scaling and costly premature RO membrane failure and frequent cleaning requirements.
Before reverse osmosis should be considered, a complete cation/anion balance is required and potential foulants must be identified.
Conclusion
Reverse Osmosis is an effective and proven technology to produce water that is suitable for many industrial applications that require demineralized or deionized water.
Proper pre-treatment and monitoring of an RO system is crucial to preventing costly repairs and unscheduled maintenance. With the correct system design, maintenance program, and experienced service support, your RO system should provide many years of high purity water.







